For more than 185,000 Americans every year, the realities of losing a limb are all too clear. For those who have to live with an amputation or a limb difference, the effects the loss of full movement can have on their health, wellbeing and self-image can be staggering. According to a survey by the Amputee Coalition, most amputees reported that their loss of limb effectively reduced their capability to perform typical tasks — with 30 percent reporting that bathing is now difficult, 7 percent reporting that routine tasks, such as eating, getting out of bed or using the bathroom being a challenge and nearly one-third of all surveyed being forced to retire due to amputation. 30 percent reported being depressed.
As African-Americans are 1.5 to 3.5 times more likely to be a candidate of amputation than Whites, and Hispanics are 3.6 times more likely to be a candidate — due in large part to diabetes and vascular disease being the leading causes for limb death — limb loss presents a greater challenge to the minority community, which statistically has less access to quality health care than the White community. Efforts to help restore quality of life to those forced to lose a part of their physical identity have driven recent efforts in prosthetics and wearable robotics.
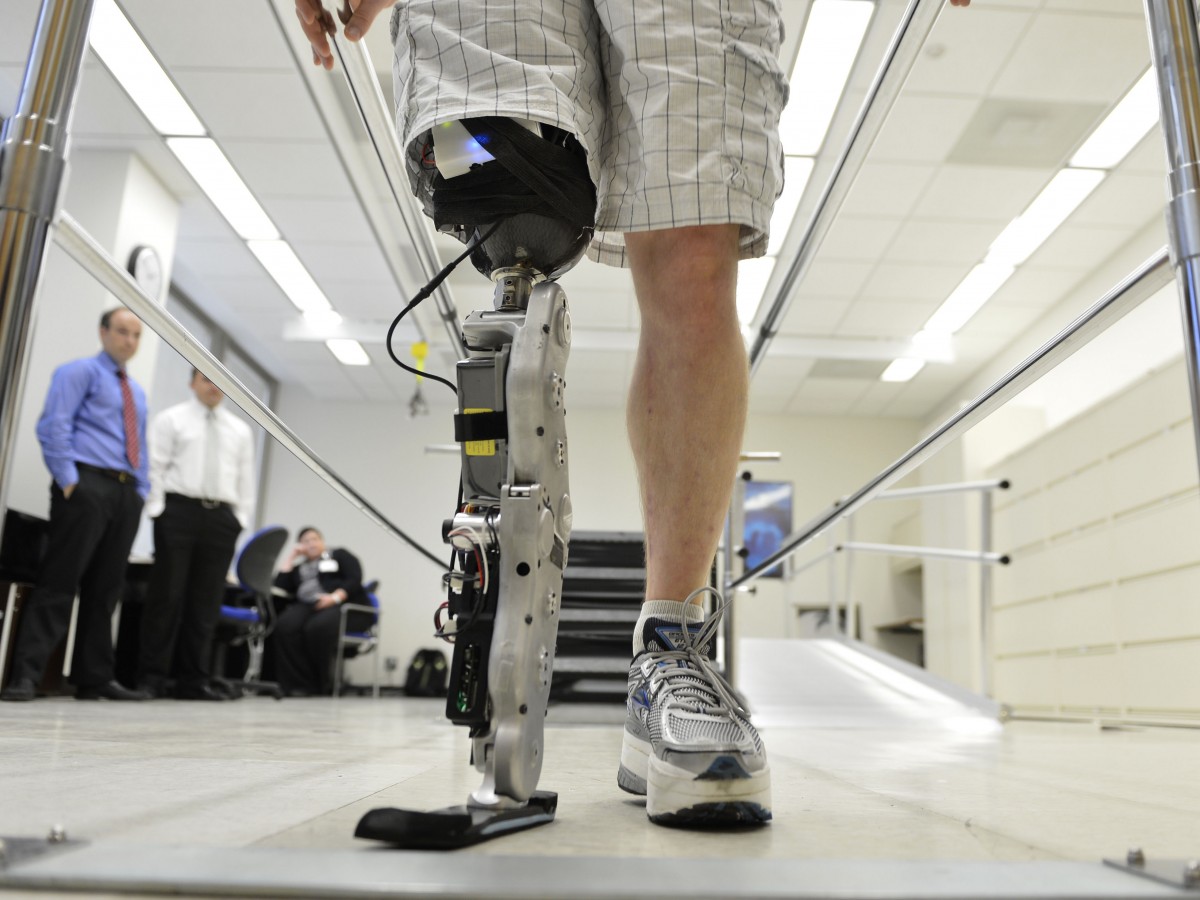
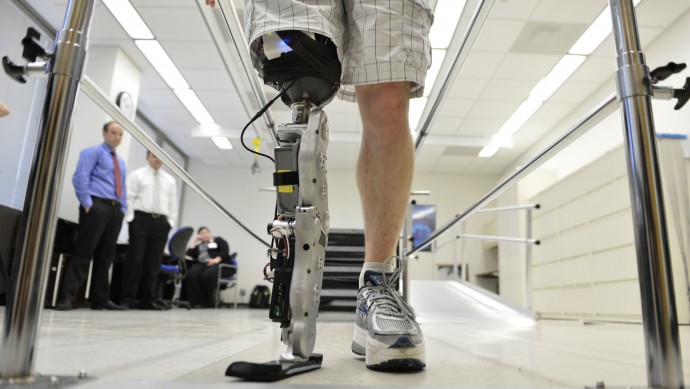
One such effort has been reported to the New England Journal of Medicine where scientists at the Rehabilitation Institute of Chicago announced the first fitting of a thought-controlled motorized limb. Based on a $8 million grant from the U.S. Army’s telemedicine and Advanced Technology Research Center, the researchers were able to produce a bionic leg that can dynamically adjust to body sway, can handle stairs and inclines and can adjust to rising from and lowering into a chair better than any other prosthetic device currently available.
“In my mind, it’s still the same thing in terms of moving my ankle down or up, or extending my leg forward or back,” said Zac Vawter, the bionic leg’s test recipient, in a telephone interview. “It’s just walk like I would normally walk. It’s not special training or buttons or tricks. That’s a big piece of what I think is groundbreaking and phenomenal about this work.” Vawter is only “field testing” the device, helping to map further refinements that will be needed before commercial release. An outside company, Freedom Innovations LLC, is working to make the prosthetic device more compact, streamlined and quieter.
“This is a groundbreaking development,” said lead author Levi Hargrove, a biomedical engineer and research scientist at RIC. “It allows people to seamlessly transition between walking along level ground and going up and down stairs and slopes.”
The mind-machine threshold
Most leg prosthetics are spring-loaded, which gives a “bounceback” whenever the wearer steps and cushions the shock the continuous impacts radiate through the body when the wearer walks. Beyond this, motorized prostheses rely on pressure pads that would measure weight distribution and muscle flex to determine how it should move. These devices require the wearer to learn a new set of movements, such as leading with the good leg or intentionally flexing the femoral muscle before taking a step. This leads to discomfort and incoordination.
Vawter, in his trials so far, has noticed that he is — for the first time since his 2009 motorcycle crash — able to walk foot-over-foot and climb stairs while alternating lead feet. “It’s still a prosthetic, but it’s in between the leg I wear every day and prior to amputation,” said Vawter, who report he can’t yet jump to the rim of a basketball net with the robotic leg, as he could before the amputation. “It’s a dramatic improvement over my current prosthetic, but there is still a long way to go.”
What makes the new prosthetic interesting is that it is directly connected to the user’s nervous system, instead of having to rely on the muscularly interpreted signals that the wearer would send a “traditional” robotic prosthetic. This reduces the error rate from 12.9 percent to 1.8 percent. The prosthesis works through “osseointegration,” a technique developed by the Chalmer University of Technology in Sweden (where the first thought-control robotic arm was developed), which grafts a metallic electrode, typically made of titanium, to living bone to create a connection point for the remaining living nerves at the amputation site and the robot.
Electrical signaling from the wearer’s brain can now be detected directly across the electrode. More importantly, electrical signaling can be fed back to the electrode, giving the wearer neural feedback similar to what he/she may experience with a natural limb. This feedback can constitute a sense of touch, which is critical in walking. Without this, the wearer would have to “guesstimate” his or her gait and foot positioning based on sight.
Beyond the limits of the flesh
Osseointegration represents the first legitimate brain to machine interface. This represents half of the challenge in creating computers capable of directly interfacing with the brain. The other part of the solution is figuring out the “neural code,” or the brain’s operating language. The brain is a complex network of neurons, which transmit data and instructions via a complicated pattern of electrical pulses. By mimicking this firing pattern, it is theoretically possible to send the brain sensory data. In the case of Vawter, the prosthetic’s software and hardware had to be tuned specifically for him, but with a broader understanding of neural coding, it may be possible to create fully artificial replacement eyes, for example.
To a certain extent, technology has progressed to the point that primitive attempts at neural coding have made digital manipulation of senses possible. The cochlear implant — an external microphone and speech processor which connects to an array of up to 22 surgically-implanted electrodes to the inner ear — have given improved hearing to more than 200,000 deaf or hard-of-hearing people worldwide. Researchers at Weill Medical College of Cornell University claimed to have figured out the neural coding of mice and monkeys and are currently optimistic that artificial receptors can be effectively connected to a patient’s optical nerve.
“We can make blind mouse retinas see, and we’re moving as fast as we can to do the same in humans,” lead researcher Sheila Nirenberg said in a statement.
In theory, this technology has the capability of making almost every part of the body — save the brain — replaceable. This presents the potential of a restoral of life and capability thought impossible and reserved for science-fiction, just a decade before.
Experts are estimating that the thought-controlled prosthetic will be available commercially between three to five years. While there is no current price attached to the prosthetic, upper extremity prosthesis can range from $20,000 to $120,000. “We are leveraging developments in related industries to make sure we use low-cost components whenever possible,” Hargrove told NBC News.
Hargrove seeks to make the device as affordable as possible toward restoring “full ability” to all patients, particularly, the elderly. “This could mean the difference between living in their home longer and having to go to a nursing home,” said Hargrove.